Chen, Popel, 2007
Model Status
This CellML model runs in OpenCell and COR and is known to be mathematically consistent with the publication it was based on. The units are consistent. An additional variable and equation has been added to allow the CellML model to reproduce Figure 2 of the publication. The three different graphs of this figure can be produced by altering the variable 'Fe3' in the 'Fe3' component (this variable represents the concentration of NOS-1 in its Fe3+ bound state) to 0.3, 0.6 or 0.9 micromolar. In this particular version of the model it has been set to 0.6.
Model Structure
ABSTRACT: Nitric oxide (NO) derived from nitric oxide synthase (NOS) is an important paracrine effector that maintains vascular tone. The release of NO mediated by NOS isozymes under various O(2) conditions critically determines the NO bioavailability in tissues. Because of experimental difficulties, there has been no direct information on how enzymatic NO production and distribution change around arterioles under various oxygen conditions. In this study, we used computational models based on the analysis of biochemical pathways of enzymatic NO synthesis and the availability of NOS isozymes to quantify the NO production by neuronal NOS (NOS1) and endothelial NOS (NOS3). We compared the catalytic activities of NOS1 and NOS3 and their sensitivities to the concentration of substrate O(2). Based on the NO release rates predicted from kinetic models, the geometric distribution of NO sources, and mass balance analysis, we predicted the NO concentration profiles around an arteriole under various O(2) conditions. The results indicated that NOS1-catalyzed NO production was significantly more sensitive to ambient O(2) concentration than that catalyzed by NOS3. Also, the high sensitivity of NOS1 catalytic activity to O(2) was associated with significantly reduced NO production and therefore NO concentrations, upon hypoxia. Moreover, the major source determining the distribution of NO was NOS1, which was abundantly expressed in the nerve fibers and mast cells close to arterioles, rather than NOS3, which was expressed in the endothelium. Finally, the perivascular NO concentration predicted by the models under conditions of normoxia was paradoxically at least an order of magnitude lower than a number of experimental measurements, suggesting a higher abundance of NOS1 or NOS3 and/or the existence of other enzymatic or nonenzymatic sources of NO in the microvasculature.
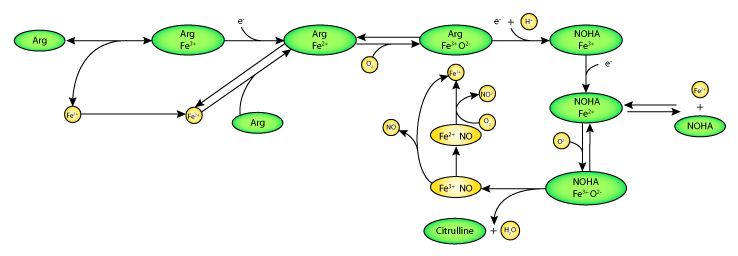 |
| Schematic diagram of the mathematical model. Pathway of NO release and transport catalysed by neuronal nitric oxide synthase ((NOS1), here represented by the heme iron (Fe)). The heme ion of NOS1 binds L-arginine (Arg), hydroxyl-L-arginine (NOHA), and oxygen (O2), and undergoes a series of oxygenation and reduction reactions. |
The original paper reference is cited below:
Vascular and perivascular nitric oxide release and transport: biochemical pathways of neuronal nitric oxide synthase (NOS1) and endothelial nitric oxide synthase (NOS3), Kejing Chen and Aleksander S. Popel, 2007, Free Radical Biology and Medicine , 42, 811-822. PubMed ID: 17320763

