Swanson, True, Lin, Buhler, Vessella, Murray, 2001
Model Status
This CellML model runs in both OpenCell and COR, the units are consistent and parameter values for constants have been set to LuCaP23.1 from table 1. The general shape of the simulation output seems correct (figure 1), however the scale is not the same as the figure in the published paper.
Model Structure
ABSTRACT: Prostate-specific antigen (PSA) is an enzyme produced by both normal and cancerous prostate epithelial cells. Although PSA is the most widely used serum marker to detect and follow patients with prostatic adenocarcinoma, there are certain anomalies in the values of serum levels of PSA that are not understood. We developed a mathematical model for the dynamics of serum levels of PSA as a function of the tumor volume. Our model results show good agreement with experimental observations and provide an explanation for the existence of significant prostatic tumor mass despite a low-serum PSA. This result can be very useful in enhancing the use of serum PSA levels as a marker for cancer growth.
The original paper is cited below:
A Quantitative Model for the Dynamics of Serum Prostate-Specific Antigen as a Marker for Cancerous Growth, Kristin R. Swanson, Lawrence D. True, Daniel W. Lin, Kent R. Buhler, Robert Vessella, and James D. Murray, 2001, American Journal of Pathology, 158, 2195-2199. PubMed ID: 11395397
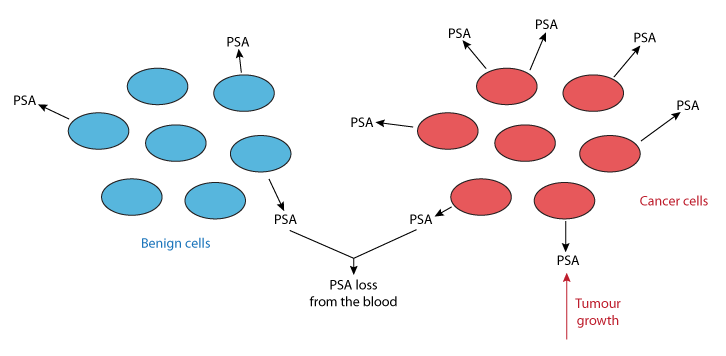 |
| The mathematical model shown visually in the above diagram can be written in words as: the rate of change of PSA equals the secretion of PSA from benign cells plus the secretion of PSA from cancerous cells minus the loss of PSA from the blood due to its metabolism and clearance. Cancer cells secrete more PSA than do benign cells. The volume of benign cells is assumed to remain constant, and the volume of cancerous cells is dependent on the size of the initial implanted tumour and the rate of tumour growth. |

