Decker, Heijman, Silva, Hund, Rudy, 2009
Model Status
This CellML model is a translation of the original MATLAB code of the original published model. The CellML model runs in both OpenCell and COR but does not yet recreate the published results - the action potential is close but the calcium dynamics still require some fixing. The units have been checked and they are consistent. We will continue to curate this model. See the investigation performed by Gary Mirams under Tracker item 3652 describing the changes introduced to improve the ability of this CellML encoding of the model to match the results from the original source code.
Model Structure
ABSTRACT: Computational models of cardiac myocytes are important tools for understanding ionic mechanisms of arrhythmia. This work presents a new model of the canine epicardial myocyte that reproduces a wide range of experimentally observed rate-dependent behaviors in cardiac cell and tissue, including action potential (AP) duration (APD) adaptation, restitution, and accommodation. Model behavior depends on updated formulations for the 4-aminopyridine-sensitive transient outward current (I(to1)), the slow component of the delayed rectifier K(+) current (I(Ks)), the L-type Ca(2+) channel current (I(Ca,L)), and the Na(+)-K(+) pump current (I(NaK)) fit to data from canine ventricular myocytes. We found that I(to1) plays a limited role in potentiating peak I(Ca,L) and sarcoplasmic reticulum Ca(2+) release for propagated APs but modulates the time course of APD restitution. I(Ks) plays an important role in APD shortening at short diastolic intervals, despite a limited role in AP repolarization at longer cycle lengths. In addition, we found that I(Ca,L) plays a critical role in APD accommodation and rate dependence of APD restitution. Ca(2+) entry via I(Ca,L) at fast rate drives increased Na(+)-Ca(2+) exchanger Ca(2+) extrusion and Na(+) entry, which in turn increases Na(+) extrusion via outward I(NaK). APD accommodation results from this increased outward I(NaK). Our simulation results provide valuable insight into the mechanistic basis of rate-dependent phenomena important for determining the heart's response to rapid and irregular pacing rates (e.g., arrhythmia). Accurate simulation of rate-dependent phenomena and increased understanding of their mechanistic basis will lead to more realistic multicellular simulations of arrhythmia and identification of molecular therapeutic targets.
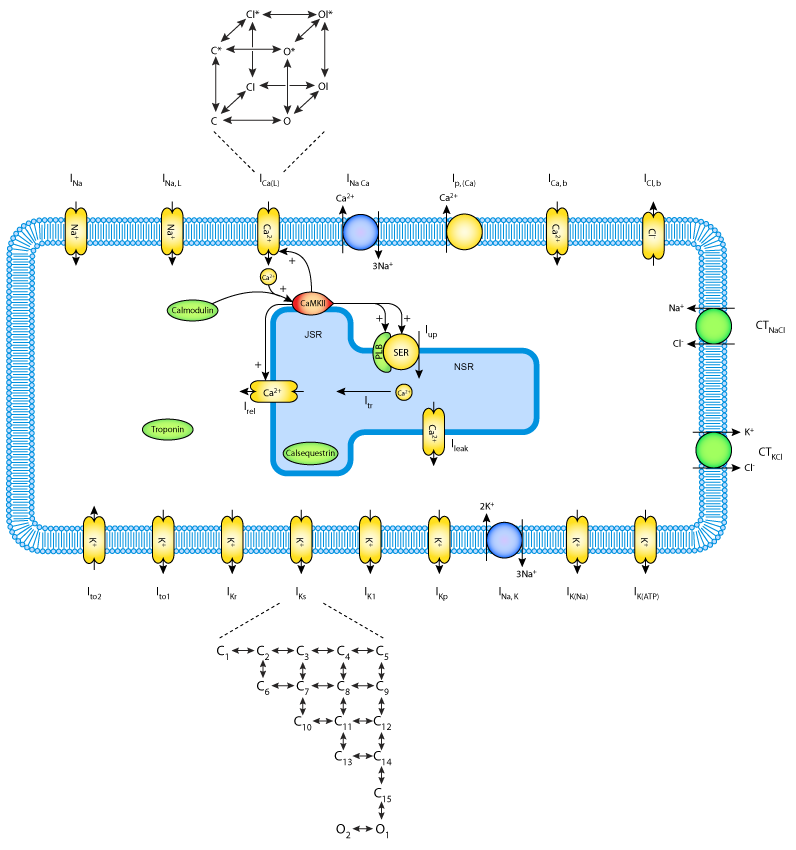 |
| Schematic diagram of the Decker at al. canine ventricular myocyte model (2009). |
The complete paper reference is cited below:
Properties and ionic mechanisms of action potential adaptation, restitution, and accommodation in canine epicardium, Keith F. Decker, Jordi Heijman, Jonathan R. Silva, Thomas J. Hund, and Yoram Rudy, 2009, American Journal of Physiology, 296, (4), H1017-H1026. PubMed ID: 19168720

