Lindblad, Murphey, Clark, Giles, 1996
Model Status
This model runs in OpenCell and COR and reproduce the published results. The units have been checked and they are consistent.
Model Structure
ABSTRACT: We have developed a mathematical model of the rabbit atrial myocyte and have used it in an examination of the ionic basis of the atrial action potential. Available biophysical data have been incorporated into the model to quantify the specific ultrastructural morphology, intracellular ion buffering, and time- and voltage-dependent currents and transport mechanisms of the rabbit atrial cell. When possible, mathematical expressions describing ionic currents identified in rabbit atrium are based on whole cell voltage-clamp data from enzymatically isolated rabbit atrial myocytes. This membrane model is coupled to equations describing Na+, K+, and Ca2+ homeostasis, including the uptake and release of Ca2+ by the sarcoplasmic reticulum and Ca2+ buffering. The resulting formulation can accurately simulate the whole cell voltage-clamp data on which it is based and provides fits to a family of rabbit atrial cell action potentials obtained at 35 degrees C over a range of stimulus rates (0.2-3.0 Hz). The model is utilized to provide a qualitative prediction of the intracellular Ca2+ concentration transient during the action potential and to illustrate the interactions between membrane currents that underlie repolarization in the rabbit atrial myocyte.
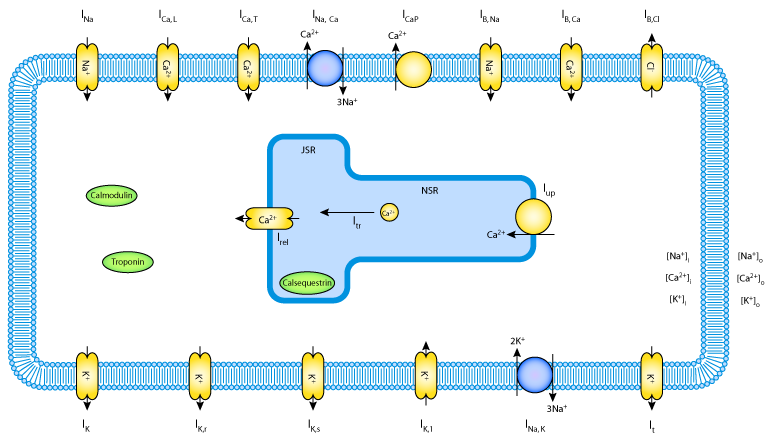 |
| A schematic representation of the Linblad et al 1996 model. |
The original paper reference is cited below:
A model of the action potential and underlying membrane currents in a rabbit atrial cell, D. S. Lindblad, C. R. Murphey, J. W. Clark and W. R. Giles, 1996, American Journal of Physiology, 271, H1666-H1696. PubMed ID: 8897964.

