Martins, Mendes, Coreiro, Freire, 2001
Model Status
This model runs in both OpenCell and COR but does not replicate the published results - probably because the publication does not define the substate HTA. The units have been checked and they are consistent.
Model Structure
ABSTRACT: The kinetics of glyoxalase I [(R)-S-lactoylglutathione methylglyoxal-lyase; EC 4.4.1.5] and glyoxalase II (S-2-hydroxyacylglutathione hydrolase; EC 3.1.2.6) from Saccharomyces cerevisiae was studied in situ, in digitonin permeabilized cells, using two different approaches: initial rate analysis and progress curves analysis. Initial rate analysis was performed by hyperbolic regression of initial rates using the program HYPERFIT. Glyoxalase I exhibited saturation kinetics on 0.05-2.5 mM hemithioacetal concentration range, with kinetic parameters Km 0.53 +/- 0.07 mM and V (3.18 +/- 0.16) x 10(-2) mM.min(-1). Glyoxalase II also showed saturation kinetics in the SD-lactoylglutathione concentration range of 0.15-3 mM and Km 0.32 +/- 0.13 mM and V (1.03 +/- 0.10) x 10(-3) mM.min(-1) were obtained. The kinetic parameters of both enzymes were also estimated by nonlinear regression of progress curves using the raw absorbance data and integrated differential rate equations with the program GEPASI. Several optimization methods were used to minimize the sum of squares of residuals. The best parameter fit for the glyoxalase I reaction was obtained with a single curve analysis, using the irreversible Michaelis-Menten model. The kinetic parameters obtained, Km 0.62 +/- 0.18 mM and V (2.86 +/- 0.01) x 10(-2) mM.min(-1), were in agreement with those obtained by initial rate analysis. The results obtained for glyoxalase II, using either the irreversible Michaelis-Menten model or a phenomenological reversible hyperbolic model, showed a high correlation of residuals with time and/or high values of standard deviation associated with Km. The possible causes for the discrepancy between data obtained from initial rate analysis and progress curve analysis, for glyoxalase II, are discussed.
The original paper reference is cited below:
In situ kinetic analysis of glyoxalase I and glyoxalase II in Saccharomyces cerevisiae, Margarida Martins, A. et al, 2001, European Journal of Biochemistry, 268, 3930-3936. PubMed ID: 11453985
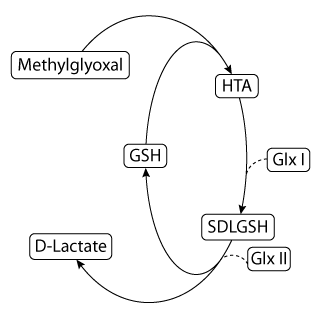 |
| A rendering of the glyoxalase pathway. Species are represented by rounded rectangles, and reactions by arrows. The action of a catalyst on a reaction is represented by dashed lines. |

