Calzone, Thieffry, Tyson, Novak, 2007
Model Status
This CellML version of the model runs in PCEnv but requires initial conditions and 'switches' in order to reproduce the published results. The set of parameters used to produce figure 3 of the published paper is implemented in this version. The doubling of N and rescaling of other variables at division are not included in this CellML description.
ValidateCellML verifies this model as valid CellML with fully consistent units.
Model Structure
ABSTRACT: Immediately following fertilization, the fruit fly embryo undergoes 13 rapid, synchronous, syncytial nuclear division cycles driven by maternal genes and proteins. During these mitotic cycles, there are barely detectable oscillations in the total level of B-type cyclins. In this paper, we propose a dynamical model for the molecular events underlying these early nuclear division cycles in Drosophila. The model distinguishes nuclear and cytoplasmic compartments of the embryo and permits exploration of a variety of rules for protein transport between the compartments. Numerical simulations reproduce the main features of wild-type mitotic cycles: patterns of protein accumulation and degradation, lengthening of later cycles, and arrest in interphase 14. The model is consistent with mutations that introduce subtle changes in the number of mitotic cycles before interphase arrest. Bifurcation analysis of the differential equations reveals the dependence of mitotic oscillations on cycle number, and how this dependence is altered by mutations. The model can be used to predict the phenotypes of novel mutations and effective ranges of the unmeasured rate constants and transport coefficients in the proposed mechanism.
The complete original paper reference is cited below:
Dynamical modeling of syncytial mitotic cycles in Drosophila embryos, Laurence Calzone, Denis Thieffry, John J Tyson, Bela Novak, 2007, Molecular Systems Biology , 3, 131. PubMed ID: 17667953
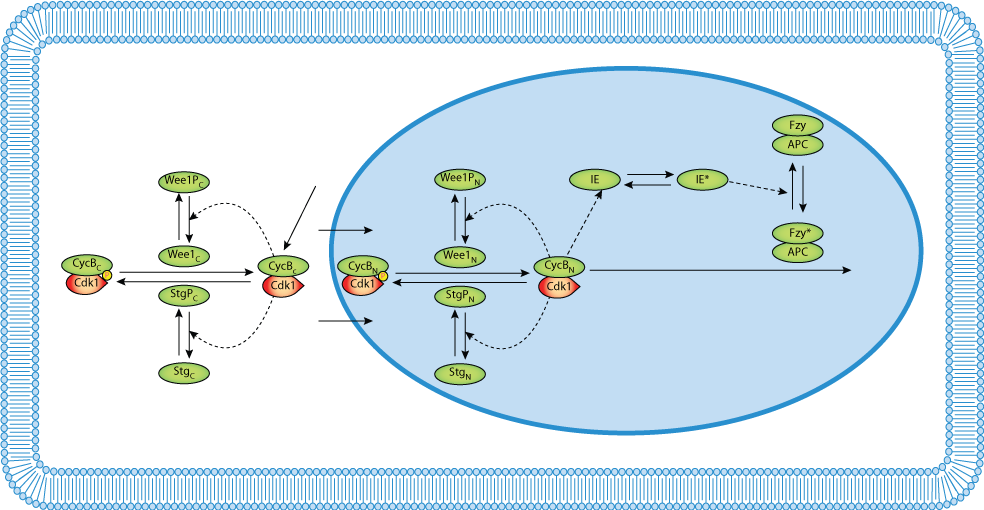 |
| Dynamical model of nuclear division cycles during early embryogenesis of Drosophila. |

