Butera, Rinzel, Smith, 1999
Model Status
This CellML model runs in OpenCell and COR to reproduce the published results (Figure 4 A3 where E_L = -57.5 mv). Please note that the model has to be run for a duration of 10000 ms with a step size of 0.01 ms and a high point density of 100000 points/graph. This model represents model 1 from the published paper (which does not include a slow potassium current).
Model Structure
ABSTRACT: A network of oscillatory bursting neurons with excitatory coupling is hypothesized to define the primary kernel for respiratory rhythm generation in the pre-Botzinger complex (pre-BotC) in mammals. Two minimal models of these neurons are proposed. In model 1, bursting arises via fast activation and slow inactivation of a persistent Na+ current INaP-h. In model 2, bursting arises via a fast-activating persistent Na+ current INaP and slow activation of a K+ current IKS. In both models, action potentials are generated via fast Na+ and K+ currents. The two models have few differences in parameters to facilitate a rigorous comparison of the two different burst-generating mechanisms. Both models are consistent with many of the dynamic features of electrophysiological recordings from pre-BotC oscillatory bursting neurons in vitro, including voltage-dependent activity modes (silence, bursting, and beating), a voltage-dependent burst frequency that can vary from 0.05 to >1 Hz, and a decaying spike frequency during bursting. These results are robust and persist across a wide range of parameter values for both models. However, the dynamics of model 1 are more consistent with experimental data in that the burst duration decreases as the baseline membrane potential is depolarized and the model has a relatively flat membrane potential trajectory during the interburst interval. We propose several experimental tests to demonstrate the validity of either model and to differentiate between the two mechanisms.
The complete original paper reference is cited below:
Models of Respiratory Rhythm Generation in the Pre-Botzinger Complex. I. Bursting Pacemaker Neurons, Robert J. Butera, Jr., John Rinzel and Jeffrey C. Smith, 1999, Journal of Neurophysiology, 81, 382-397. PubMed ID: 10400966
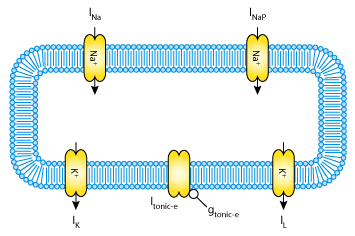 |
| The first mathematical model is based on a single-compartment Hodgkin-Huxley type formalism. It is composed of five ionic currents across the plasma membrane: a fast sodium current, INa; a delayed rectifier potassium current, IK; a persistent sodium current, INaP; a passive leakage current, IL; and a tonic current, Itonic_e (although this last current is considered to be inactive in these models). |
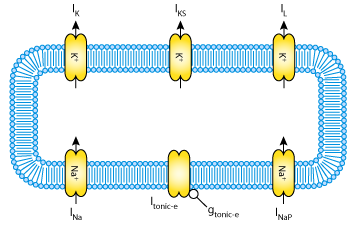 |
| The second model appears identical to the first except with the addition of a slow K+ current, IKS. (The removal of the inactivation term "h" from INaP is not visible in the model diagram.) |

