Demir, Clark, Giles, 1999
Model Status
This model has been validated by Penny Noble of Oxford University and is known to run in COR and OpenCell to reproduce the published results. The units have been checked and are balanced.
Model Structure
ABSTRACT: We have extended our compartmental model [Am. J. Physiol. 266 (Cell Physiol. 35): C832-C852, 1994] of the single rabbit sinoatrial node (SAN) cell so that it can simulate cellular responses to bath applications of ACh and isoprenaline as well as the effects of neuronally released ACh. The model employs three different types of muscarinic receptors to explain the variety of responses observed in mammalian cardiac pacemaking cells subjected to vagal stimulation. The response of greatest interest is the ACh-sensitive change in cycle length that is not accompanied by a change in action potential duration or repolarization or hyperpolarization of the maximum diastolic potential. In this case, an ACh-sensitive K+ current is not involved. Membrane hyperpolarization occurs in response to much higher levels of vagal stimulation, and this response is also mimicked by the model. Here, an ACh-sensitive K+ current is involved. The well-known phase-resetting response of the SAN cell to single and periodically applied vagal bursts of impulses is also simulated in the presence and absence of the beta-agonist isoprenaline. Finally, the responses of the SAN cell to longer continuous trains of periodic vagal stimulation are simulated, and this can result in the complete cessation of pacemaking. Therefore, this model is 1) applicable over the full range of intensity and pattern of vagal input and 2) can offer biophysically based explanations for many of the phenomena associated with the autonomic control of cardiac pacemaking.
The original paper reference is cited below:
Parasympathetic modulation of sinoatrial node pacemaker activity in rabbit heart: a unifying model, Semahat S. Demir, John W. Clark and Wayne R. Giles, 1999, American Journal of Physiology, 276, H2221-H2244. PubMed ID: 10362707
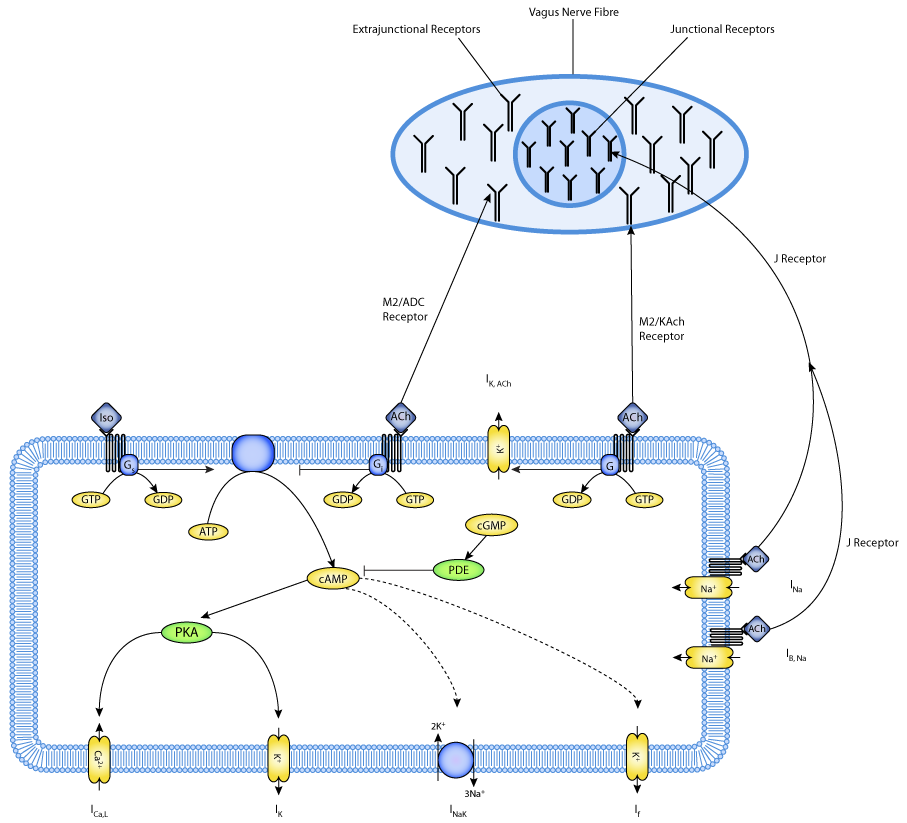 |
| A schematic diagram describing the current flows across the cell membrane that are captured in the Demir et al 1999 model of the electrophysiological activity in a SAN cell. |

