Kongas, van Beek, 2001
Model Status
This CellML model is known to run on both OpenCell and COR. The units have been checked and they are balanced. The figures in the original publication display slightly unusual variables, hence we are unsure as to whether or not the CellML model is recreating the published results. To address this we are working with the original model authors to complete the curation of the CellML model. Please also note there are some typographical errors in the original publication, and that in equations 6-10 in the appendix, the metabolites should have an "i" subscript(to denote IMS concentrations), and also the "Vhyd"s in these equations should be "Vsyn"s.
Model Structure
ATP is the major energy currency in cells. It is synthesised during aerobic respiration in the mitochondria and it's main use in muscle is in myofibrillar contraction an ion pumping. Phosphocreatine (PCr) is another major energy carrier in muscle cells, in fact the majority of the energy-carrying phosphate groups leave the mitochondria in the form of PCr, not ATP. This is possible due to the creatine kinase (CK) reaction, see Figure 1 below, where Cr is creatine:
 |
| Figure 1. The creatine kinase reaction. |
The mitochondrial isoform of the CK enzyme (MiCK) produces PCr, while the main myofibrillar isoform (MMCK) converts it back into ATP. This energy transfer is also referred to as the phosphocreatine shuttle, and it facilitates energy transfer by providing a parallel pathway to ATP diffusion. In muscle cells, the CK system also acts as an energy buffer, allowing ATP levels to remain fairly constant during muscle contraction as PCr levels fluctuate to closely follow force generation.
In the Kongas and van Beek 2001 publication described here, the authors aim to determine the mode of functioning of the CK system in the heart. To this end they devloped a mathematical model of energy production, transfer, and consumption (see Figure 2 below) and analysed the roles of different CK isoenzymes in the dynamic response to workload transitions.
The original paper reference is cited below:
Creatine kinase in energy metabolic signaling in muscle, Olav Kongas and Johannes H. G. M. van Beek, 2001, ICSB2001: The 2nd International Conference on Systems Biology
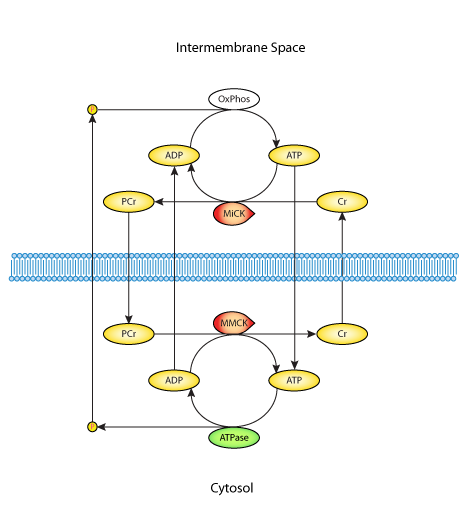 |
| Figure 2. Schematic diagram of the mathematical model. The model contains two compartments: the cytosol and the intermembrane space, and these are separated by a partialy permeable membrane (dashed line). Vertical arrows across this membrane represent diffusional fluxes. The horizontal arrows represent reactions, namely ATP synthesis (OxPhos), ATP hydrolysis (ATPase), mitochondrial and myofibrillar kinases (MiCK and MMCK). |

