Chang, Fujita, 2001
Model Status
This CellML model is a description of Chang and Fujita's 2001 mathematical model of a H-ATPase in the distal tubule of the rat: it is one component of an overall model of acid/base transport in a distal tubule.
Model Structure
ABSTRACT: The purpose of this study is to develop a numerical model that simulates acid-base transport in rat distal tubule. We have previously reported a model that deals with transport of Na(+), K(+), Cl(-), and water in this nephron segment (Chang H and Fujita T. Am J Physiol Renal Physiol 276: F931-F951, 1999). In this study, we extend our previous model by incorporating buffer systems, new cell types, and new transport mechanisms. Specifically, the model incorporates bicarbonate, ammonium, and phosphate buffer systems; has cell types corresponding to intercalated cells; and includes the Na/H exchanger, H-ATPase, and anion exchanger. Incorporation of buffer systems has required the following modifications of model equations: new model equations are introduced to represent chemical equilibria of buffer partners [e.g., pH = pK(a) + log(10) (NH(3)/NH(4))], and the formulation of mass conservation is extended to take into account interconversion of buffer partners. Furthermore, finite rates of H(2)CO(3)-CO(2) interconversion are taken into account in modeling the bicarbonate buffer system. Owing to this treatment, the model can simulate the development of disequilibrium pH in the distal tubular fluid. For each new transporter, a state diagram has been constructed to simulate its transport kinetics. With appropriate assignment of maximal transport rates for individual transporters, the model predictions are in agreement with free-flow micropuncture experiments in terms of HCO reabsorption rate in the normal state as well as under the high bicarbonate load. Although the model cannot simulate all of the microperfusion experiments, especially those that showed a flow-dependent increase in HCO reabsorption, the model is consistent with those microperfusion experiments that showed HCO reabsorption rates similar to those in the free-flow micropuncture experiments. We conclude that it is possible to develop a numerical model of the rat distal tubule that simulates acid-base transport, as well as basic solute and water transport, on the basis of tubular geometry, physical principles, and transporter kinetics. Such a model would provide a useful means of integrating detailed kinetic properties of transporters and predicting macroscopic transport characteristics of this nephron segment under physiological and pathophysiological settings.
The original paper reference is cited below:
A numerical model of acid-base transport in rat distal tubule, Hangil Chang and Toshiro Fujita, 2001, American Journal of Physiology, 281, F222-F243. PubMed ID: 11457714.
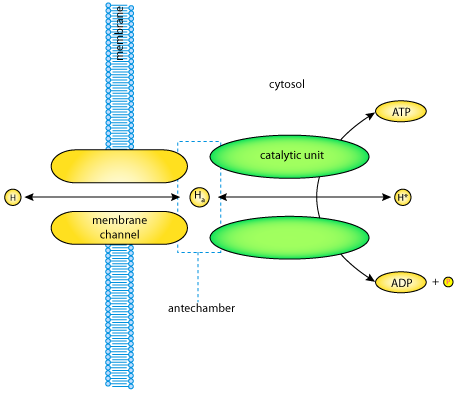 |
| Conceptual diagram of the H-ATPase. The transporter consists of two components: a transmembrane channel and an intracellular catalytic unit. Between these two components there is a buffer space known as the antechamber, in which hydrogen ions (Ha) are in equilibrium with extracellular hydrogen ions (H) due to a large conductance of the transmembrane channel. Hydrogen ions are also moved between the antechamber and the cytosol via the catalytic unit. This ion transport is coupled to ATP hydrolysis/synthesis. |
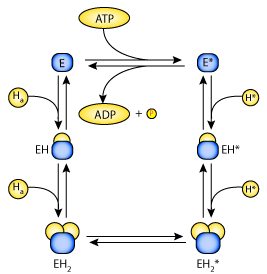 |
| State diagram of the catalytic unit of the H-ATPase. The catalytic unit (E) has two binding sites for H. Symbols with the asterisk (*) indicate conformations of the catalytic unit in which the binding sites face the cytosol, and symbols without the asterisk represent conformations in which the binding sites face the antechamber. Transition between the unloaded conformations is coupled with ATP synthesis/hydrolysis. |

