Ralser, Lehrach, Krobitsch, Wamelink, Kowald, Gerisch, Heeren, Struys, Klipp, Jakobs, Breitenbach, 2007
Model Status
This CellML model runs in COR and OpenCell and the units are consistent throughout. It reproduces the published results and was converted from SBML with the help of Lukas Endler.
Model Structure
ABSTRACT: BACKGROUND: Eukaryotic cells have evolved various response mechanisms to counteract the deleterious consequences of oxidative stress. Among these processes, metabolic alterations seem to play an important role. RESULTS: We recently discovered that yeast cells with reduced activity of the key glycolytic enzyme triosephosphate isomerase exhibit an increased resistance to the thiol-oxidizing reagent diamide. Here we show that this phenotype is conserved in Caenorhabditis elegans and that the underlying mechanism is based on a redirection of the metabolic flux from glycolysis to the pentose phosphate pathway, altering the redox equilibrium of the cytoplasmic NADP(H) pool. Remarkably, another key glycolytic enzyme, glyceraldehyde-3-phosphate dehydrogenase (GAPDH), is known to be inactivated in response to various oxidant treatments, and we show that this provokes a similar redirection of the metabolic flux. CONCLUSION: The naturally occurring inactivation of GAPDH functions as a metabolic switch for rerouting the carbohydrate flux to counteract oxidative stress. As a consequence, altering the homoeostasis of cytoplasmic metabolites is a fundamental mechanism for balancing the redox state of eukaryotic cells under stress conditions.
The original paper reference is cited below:
Dynamic rerouting of the carbohydrate flux is key to counteracting oxidative stress, Markus Ralser, Mirjam M Wamelink, Axel Kowald, Birgit Gerisch, Gino Heeren, Eduard A Struys, Edda Klipp, Cornelis Jakobs, Michael Breitenbach, Hans Lehrach and Sylvia Krobitsch, 2007, Journal of Biology, volume 6, 1-18. PubMed ID: 18154684
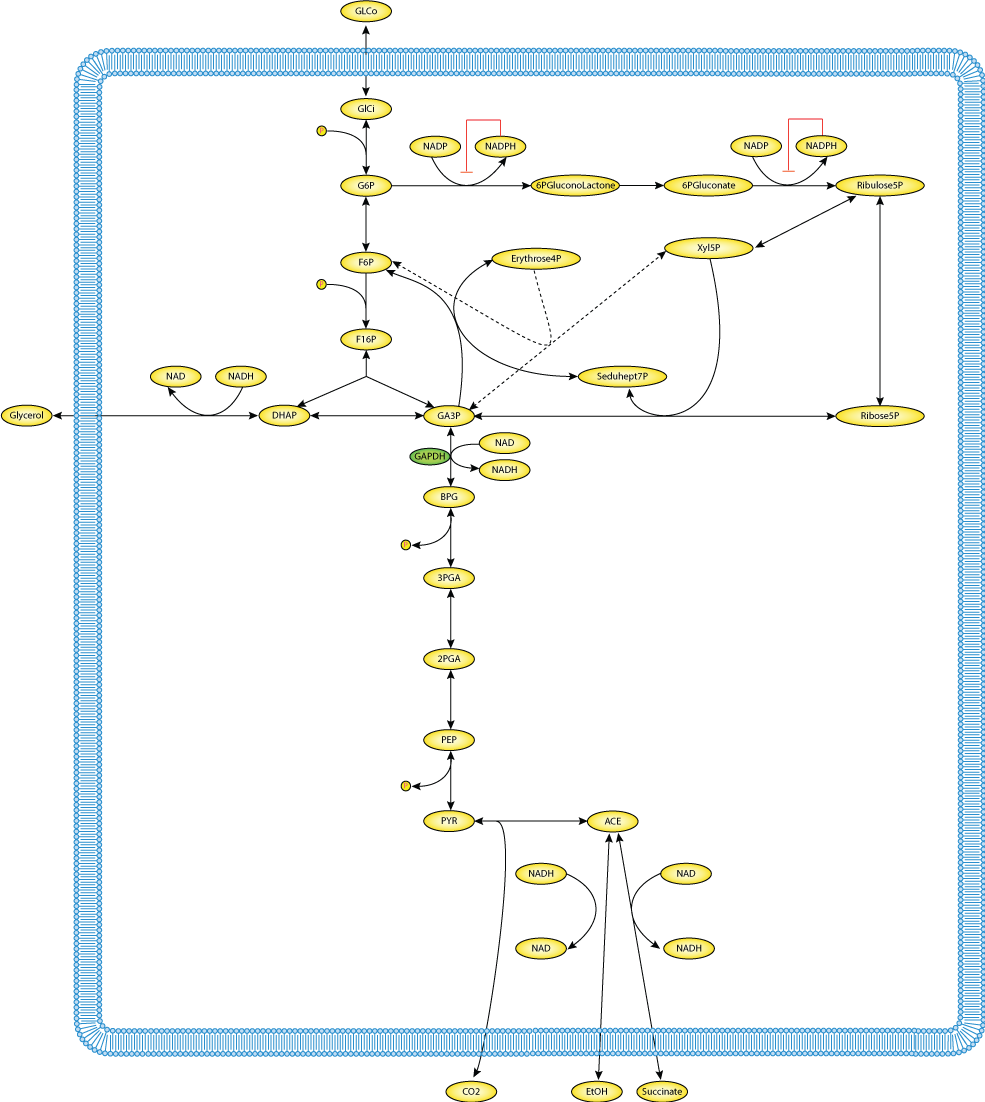 |
| Schematic diagram of the reactions included in the mathematical model to study the effects of a diminished TPI or GAPDH activity on the flux through glycolysis and the pentose phosphate pathway. Substances with a dashed outline are assumed to have a constant concentration |

