Bonhoeffer, Rembiszewski, Ortiz, Nixon, 2000
Model Status
This CellML model runs in OpenCell and COR to replicate the published results (figure 2 - up until the introduction of the drug, which is not described in this set of equations). The units have been checked and they are consistent. This particular CellML model represents model 1 from the published paper.
Model Structure
ABSTRACT: BACKGROUND: Structured interruptions of antiretroviral therapy of HIV-1 infected individuals are currently being tested in clinical trials to study the effect interruptions have on the immune responses and control of virus replication. OBJECTIVE: To investigate the potential risks and benefits of interrupted therapy using standard population dynamical models of HIV replication kinetics. METHODS: Standard population dynamical models were used to study the effect of structured therapy interruptions on the immune effector cells, the latent cell compartment and the emergence of drug resistance. CONCLUSIONS: The models suggest that structured therapy interruption only leads to transient or sustained virus control if the immune effector cells increase during therapy. This increase must more than counterbalance the increase in susceptible target cells induced by therapy. The risk of inducing drug resistance by therapy interruptions or the risk of repopulating the pool of latent cells during drug-free periods may be small if the virus population remains at levels considerably below baseline. However, if the virus load increases during drug-free periods to levels similar to or higher than baseline before therapy, both these risks increase dramatically.
The original paper reference is cited below:
Risks and benefits of structured antiretroviral drug therapy interruptions in HIV-1 infection, Sebastian Bonhoeffer, Michal Rembiszewski, Gabriel M. Ortiz, and Douglas F. Nixon, 2000, AIDS, 14, 2313-2322. PubMed ID: 11089619
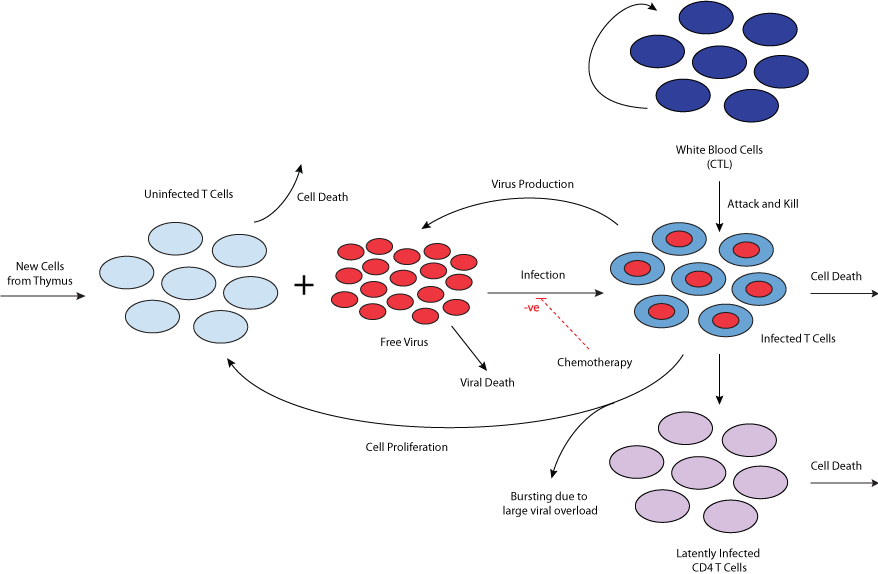 |
| Schematic diagram of a mathematical model of the interaction between HIV and the immune system. |

