Cloutier, Wellstead, 2009
Model Status
This CellML model represents version A of the published model (Table 1.A vPFK regulation by ATP-AMP concentrations) and runs in both PCEnv and COR to replicate the published results (Figure 2A). The units have been checked and they are consistent. We'd like to thank the original model author Mathieu Cloutier for his time spent curating the CellML model to get it matching the published model.
Model Structure
ABSTRACT: The biochemical regulation of energy metabolism (EM) allows cells to modulate their energetic output depending on available substrates and requirements. To this end, numerous biomolecular mechanisms exist that allow the sensing of the energetic state and corresponding adjustment of enzymatic reaction rates. This regulation is known to induce dynamic systems properties such as oscillations or perfect adaptation. Although the various mechanisms of energy regulation have been studied in detail from many angles at the experimental and theoretical levels, no framework is available for the systematic analysis of EM from a control systems perspective. In this study, we have used principles well known in control to clarify the basic system features that govern EM. The major result is a subdivision of the biomolecular mechanisms of energy regulation in terms of widely used engineering control mechanisms: proportional, integral, derivative control, and structures: feedback, cascade and feed-forward control. Evidence for each mechanism and structure is demonstrated and the implications for systems properties are shown through simulations. As the equivalence between biological systems and control components presented here is generic, it is also hypothesized that our work could eventually have an applicability that is much wider than the focus of the current study.
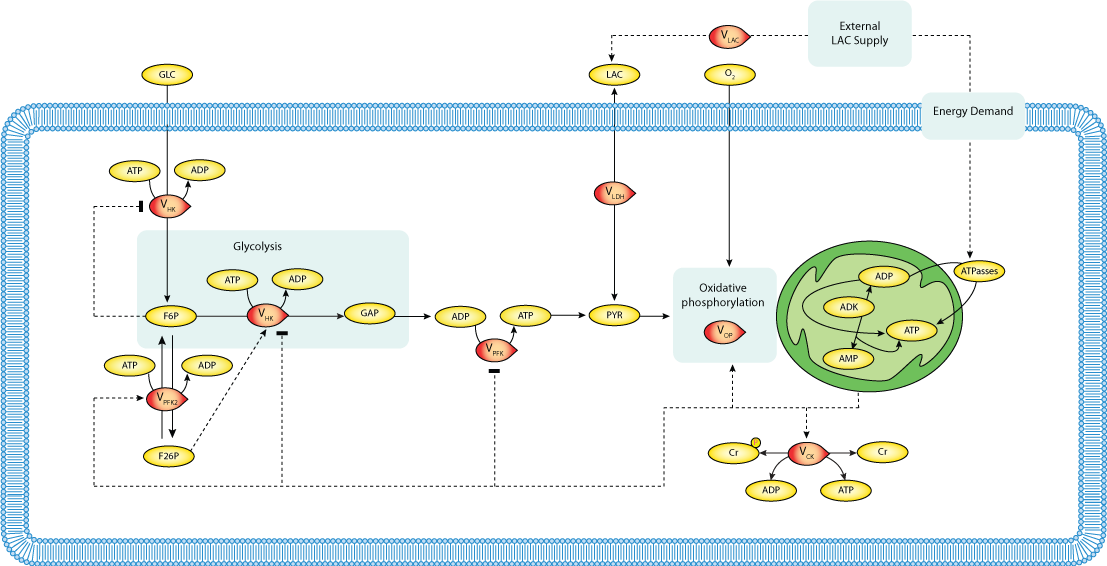 |
| Schematic diagram of a generic model for energy metabolism |
The original paper reference is cited below:
The control systems structures of energy metabolism, Mathieu Cloutier and Peter Wellstead, 2009, Journal of the Royal Society Interface, DOI: 10.1098/rsif.2009.0371. PubMed ID: 19828503

